That means they can absorb a wider range of wavelengths - including infrared waves. But greenhouse gases like CO2 and methane are made up of three or more atoms, which gives them a larger variety of ways to stretch and bend and twist. Smerdon says that the reason why some molecules absorb infrared waves and some don’t “depends on their geometry and their composition.” He explained that oxygen and nitrogen molecules are simple - they’re each made up of only two atoms of the same element - which narrows their movements and the variety of wavelengths they can interact with. Those ranges don’t overlap, so to oxygen and nitrogen, it’s as if the infrared waves don’t even exist they let the waves (and heat) pass freely through the atmosphere.īy measuring the wavelengths of infrared radiation that reaches the surface, scientists know that carbon dioxide, ozone, and methane are significantly contributing to rising global temperatures. For example, oxygen and nitrogen absorb energy that has tightly packed wavelengths of around 200 nanometers or less, whereas infrared energy travels at wider and lazier wavelengths of 700 to 1,000,000 nanometers. That’s because molecules are picky about the range of wavelengths that they interact with, Smerdon explained. Oxygen and nitrogen don’t interfere with infrared waves in the atmosphere. (Hold your hand over a dark rock on a warm sunny day and you can feel this phenomenon for yourself.) These infrared waves travel up into the atmosphere and will escape back into space if unimpeded. When sunlight reaches Earth, the surface absorbs some of the light’s energy and reradiates it as infrared waves, which we feel as heat. Credit: A loose necktie on Wikimedia Commons Greenhouse gases like carbon dioxide and methane absorb the infrared energy, re-emitting some of it back toward Earth and some of it out into space. The inadequacy of the simple Lewis structure view of molecules led to the development of the more generally applicable and accurate valence bond theory of Slater, Pauling, et al., and henceforth the molecular orbital theory developed by Mulliken and Hund.Simplified diagram showing how Earth transforms sunlight into infrared energy. In reality, the distribution of electrons in the molecule lies somewhere between these two extremes. Oxidation states overemphasize the ionic nature of the bonding the difference in electronegativity between carbon and oxygen is insufficient to regard the bonds as being ionic in nature. The oxidation state view of the CO 2 molecule is shown below: With the oxidation state formalism, the electrons in the bonds are "awarded" to the atom with the greater electronegativity. This can be most effectively visualized in an electrostatic potential map. The covalent (sharing) aspect of the bonding is overemphasized in the use of formal charges, since in reality there is a higher electron density around the oxygen atoms due to their higher electronegativity compared to the carbon atom. The formal charge view of the CO 2 molecule is essentially shown below: With formal charge, the electrons in each covalent bond are assumed to be split exactly evenly between the two atoms in the bond (hence the dividing by two in the method described above).  
The reason for the difference between these values is that formal charges and oxidation states represent fundamentally different ways of looking at the distribution of electrons amongst the atoms in the molecule. If the formal charges and oxidation states of the atoms in carbon dioxide are compared, the following values are arrived at: The concept of oxidation states constitutes a competing method to assess the distribution of electrons in molecules. 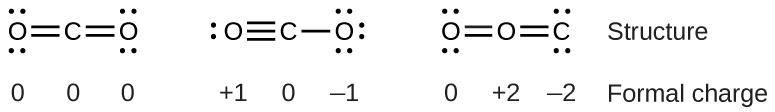
\)įormal charge compared to oxidation stateįormal charge is a tool for estimating the distribution of electric charge within a molecule.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
